This article examines the mechanism of action of peptide analogs of glucagon-like peptide-1 (GLP-1), specifically semaglutide, and the physiological consequences of their mass consumption outside of clinical protocols. We analyze the risks of receptor atrophy and gastrointestinal complications against the backdrop of current usage statistics in the USA and EU through 2026.
The Biochemical Mechanism of Satiety and Secretion
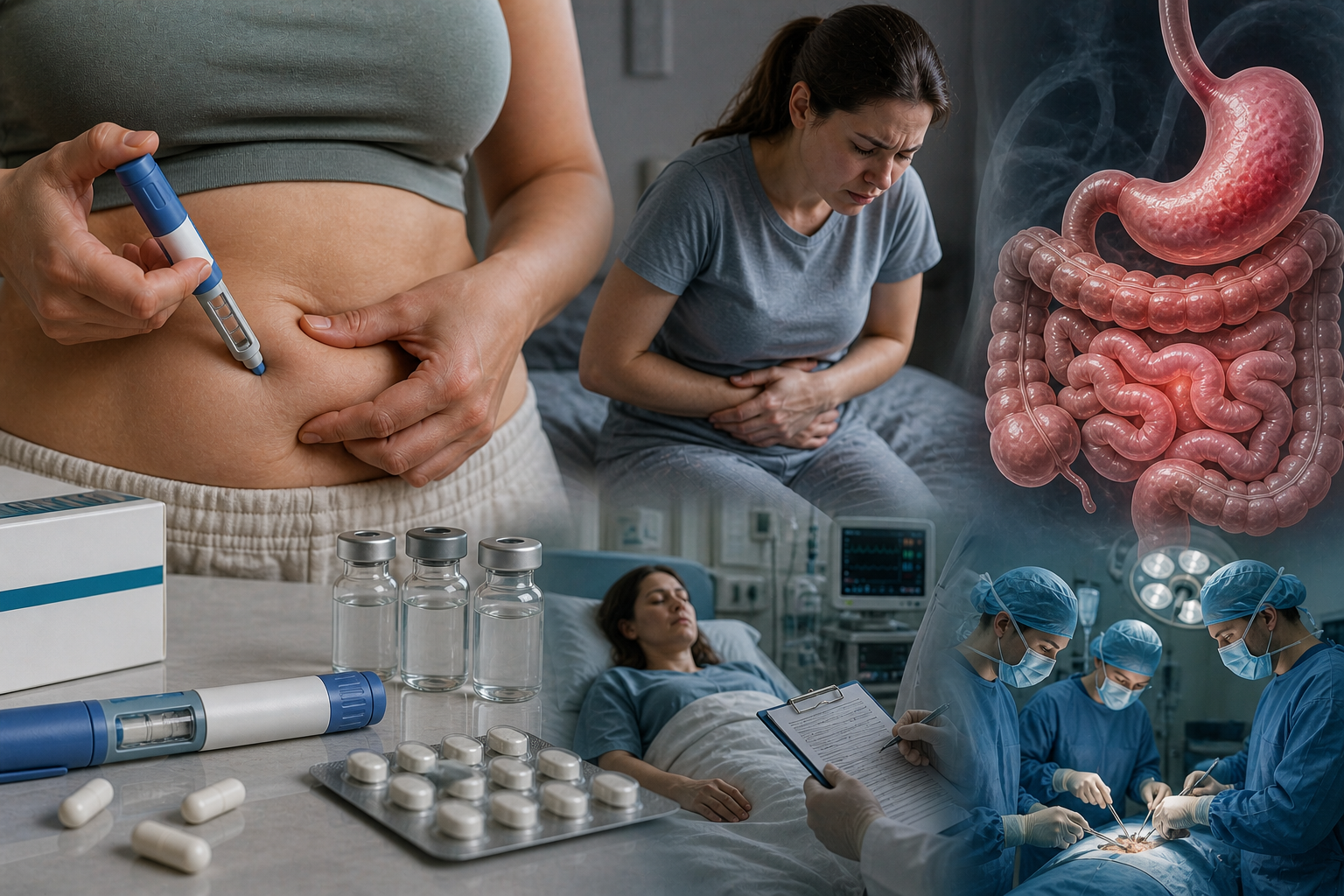
Glucagon-like peptide-1 (GLP-1) is an incretin hormone central to glucose homeostasis and appetite regulation. Its pharmacological analogs were originally engineered for Type 2 diabetes therapy but have demonstrated unprecedented efficacy in obesity correction. These peptides function as agonists of GLP-1 receptors (GLP-1R), stimulating insulin secretion while suppressing glucagon release. The primary weight-loss mechanism involves profound gastroparesis, which is the slowing of gastric emptying and a reduction in gastric acid secretion. Simultaneously, these peptides act on the arcuate nucleus of the hypothalamus to provide prolonged satiety and diminish the hedonic drive to consume food.
The Physiological Hazards of Long-Term Unsupervised Use
The “cosmetic” use of these peptides without medical supervision presents specific physiological risks that are often overlooked by the general public. Persistent stimulation of GLP-1 receptors by exogenous agonists with long half-lives can lead to receptor desensitization. In experimental models, this manifests as a “down-regulation” mechanism—a reduction in the density of active receptors on cell surfaces. This potential for functional atrophy of the incretin system suggests a dangerous dependency. Upon cessation of therapy, natural GLP-1 may no longer effectively bind to its receptors, leading to rapid weight regain and severe metabolic disturbances.
Furthermore, chronic inhibition of gastric motility can lead to permanent gastrointestinal dysfunction. Recent clinical data through 2025 and 2026 have confirmed a heightened correlation between unmonitored GLP-1R use and the development of stomach paralysis, pancreatitis, and intestinal obstruction. These conditions often arise from the tonic inhibition of the smooth muscles of the intestine, turning a metabolic aid into a chronic illness.
Global Usage Trends and Statistical Analysis (2024–2026)
By the beginning of 2026, the global market for GLP-1 agonists reached approximately $66.38 billion. The transition from clinical necessity to aesthetic demand is clearly reflected in the current regional data regarding unsupervised access.
| Region | Estimated Users (2026) | Non-prescription / Off-label Use Rate |
| United States | ~20 Million | 25% (250% increase since 2018) |
| European Union | ~12 Million | 18% (Primarily via online pharmacies) |
While the 35–64 age bracket remains the largest demographic, the 18–34 category has seen the most aggressive growth. In this younger group, usage for aesthetic purposes rather than metabolic necessity has surged, often bypassing clinical screening. This shift has led to an increase in reported adverse cases. For instance, FDA and EMA registries in 2025 documented a 34-year-old female with terminal-stage gastroparesis following 14 months of self-administered high-dose semaglutide. Similarly, in early 2026, French authorities reported over 50 cases of severe hypoglycemic coma linked to counterfeit peptides purchased online for “fast weight loss.”
Final Considerations on Public Health
Peptide analogs of GLP-1 represent a revolutionary tool in the fight against metabolic epidemics, but their transformation into “lifestyle drugs” creates a significant threat to public health. The absence of professional oversight regarding dosage titration and therapy duration risks systemic GI dysfunction and irreversible degradation of the body’s natural receptor systems. Strengthening regulatory oversight and increasing patient awareness regarding long-term physiological consequences is essential to prevent a secondary epidemic of chronic gastrointestinal paralysis.